All published articles of this journal are available on ScienceDirect.
Extinction Delay and Reinstated Fear Generalization
Abstract
Background:
After classical conditioning and extinction of fear, presentation of an unconditioned stimulus can produce a re-emergence of the extinguished fear response, a reinstatement effect. Previous research has reported that reinstatement of fear of contextual cues after a post-extinction delay could generalize over time to similar contexts.
Objective:
Two experiments were designed to investigate reinstated fear to a discrete stimulus with either a post-reinstatement delay (Experiment 1) or a post-extinction delay (Experiment 2).
Methods:
In Experiment 1 rat subjects were randomly assigned to four groups which received fear conditioning and extinction with a 3000 Hz tone CS. Reinstatement conditioning was then conducted with either the same or different CS, and testing for reinstated fear with the original CS was conducted after either a short or long delay following reinstatement. In Experiment 2 all procedures were the same except the long or short delay employed was between extinction and reinstatement instead of after reinstatement.
Results:
Reinstated fear increased with time to the same CS, and an increase in generalization of reinstated fear over time was observed with a different reinstatement CS (Experiment 1). When the delay employed was between extinction and reinstatement no increase in fear generalization was observed (Experiment 2).
Conclusion:
The results indicated support for the notion that reinstated fear to a discrete stimulus may generalize over time, but that generalization only takes place when reinstatement is administered a short interval after extinction. The results have implications for clinical behavior therapy treatments of anxiety disorders.
1. INTRODUCTION
Fear is generally defined as phasic, apprehensive arousal to an explicit threat of an aversive stimulus [1]. Although fear is generally adaptive, it may become a source of pathology when experienced in the absence of direct threat (i.e., anxiety), and anxiety disorders are among the most prevalent mental health diagnoses, reported to affect 18% of the adult population [2]. Behavioral exposure therapy, which has been a common treatment method for anxiety disorders [3, 4], involves the presentation of anxiety-producing stimuli in the absence of an aversive stimulus. While exposure treatments are overall effective in reducing fear, relapse following treatment is common. For example, Craske [5] reports that relapse rates can range between 19-62% of patients treated with this therapy method. It is therefore important to empirically examine the mechanisms underlying exposure treatments and variables involved in relapse.
It has long been recognized that behavioral learning principles, specifically classical conditioning, are involved in the development of anxiety disorders as well as the successful elimination of such fear responses e.g., [6], and laboratory research has been conducted with human [7] and infrahuman [8] subjects. In a typical conditioning situation, a neutral stimulus (conditioned stimulus, CS) is paired with an aversive stimulus (unconditioned stimulus, US), and after those pairings, a conditioned response (CR) of fear may be developed to the stimulus. If that CS is then repeatedly presented alone without the US, extinction of the fear response begins to occur, and with sufficient stimulus-alone presentations, the fear response will no longer be observable in behavior. Although responding to a CS may be absent after an extinction treatment, several post-extinction phenomena suggest that extinction does not completely erase the original learning and that some associative strength remains. For example, presenting the CS after a post-extinction delay without any further conditioning results in a reappearance of the CR (spontaneous recovery) [9, 10]), presentation of two extinguished CSs in summation may result in new responding [11], and administering extinction in a different context than training or testing can produce a renewal of the CR [12-14].
One additional phenomenon of post-extinction recovery of fear is the so-called reinstatement effect, in which a single presentation of the US after extinction can produce recovered conditioned responses [15]. Animal research on this phenomenon has attributed the reinstatement effect as a result of the US serving as a reminder cue of original conditioning [16] or to the conditioning of stimuli present when the reinstatement US is presented, such as the contextual cues [8, 14, 17]. Callen et al. [8] have suggested that the associative properties producing reinstatement may be similar to those underlying rapid reacquisition, in which additional CS-US pairings after extinction produce faster reacquisition of the CR compared to original conditioning [18]. Laboratory studies have also demonstrated reinstatement with human anxiety responses [19, 20].
The effectiveness of a reinstatement treatment to produce recovered fear has been demonstrated to be influenced by temporal factors in several studies of context conditioning. For instance, MacArdy and Riccio [16] manipulated the interval between the extinction of a conditioned fear response and administration of a noncontingent footshock in a context different than training. With a short delay (one day) between extinction and the post-extinction shock, no context fear reinstatement was observed. However, when the shock was given after a long delay (seven days after extinction), reinstatement of context fear was observed [21]. These results can be viewed as consistent with an increase in stimulus generalization over time of the reinstatement of contextual fear. Presumably, with a short delay after extinction, subjects were able to discriminate between the training context and a different post-extinction shock context. However, a longer delay allowed for more generalization of the context stimuli or a flattening of the stimulus generalization gradient over time [22].
The MacArdy and Riccio [16] research, as well as that by McAllister and McAllister [21], examined increased generalization over time of reinstated context fear. However, no research has been reported examining reinstated fear generalization over time with a discrete CS. The purpose of the present research was to use an animal model in two separate experiments to examine whether any increase in generalization of fear to a specific discrete auditory stimulus (tone) would occur as a function of time between reinstatement and fear testing (Exp. 1) as well as between extinction and reinstatement (Exp. 2).
2. MATERIAL AND METHODS
2.1. Experiment 1
2.1.1. Subjects and Design
The subjects were 40 naive albino Sprague Dawley rats, approximately 150 days old, supplied by the University of South Carolina Aiken Psychology Department animal breeding colony. All subjects were housed individually and allowed ad libitum access to food and water throughout the duration of the experiment. All experimental sessions occurred during the light cycle and were conducted at approximately the same time each day. All animal care and procedures followed were in accordance with the standards set forth in the eighth edition of “Guide for the Care and Use of Laboratory Animals,” and approval of the experimental protocol and animal care was obtained by the University of South Carolina Aiken Institutional Animal Care and Use Committee (IACUC Approval # 100609-PSY-02) prior to data collection.
Rats were randomly assigned into groups using a 2 x 2 factorial design, with Post-Reinstatement Delay (Short, Long) x Reinstatement CS (Same, Diff), yielding 4 groups: Short-Same, Short-Diff, Long-Same, Long-Diff.
2.1.2. Apparatus
Treatments were administered in four identical Med Associates operant chambers (Env-022MD). Background illumination was provided by an incandescent light bulb (28-V) centered on the left wall and mounted 16.67 cm from the floor. A speaker on the back left wall provided the 80db auditory stimuli (3000 Hz tone or 1000 Hz tone). Footshock (1 mA, 0.7 s) was delivered through the grid floor. The chambers were connected to a computer via a Med Associates Interface through which all programming and data collection were performed. A Fuji Fish Eye camera, mounted on the left front door 30 cm from the floor, was used to record and analyze freezing behavior during test trials. All chambers were housed in sound and light attenuating chambers.
2.1.3. Procedure
The general procedure consisted of five phases: Handling/Exploration, Fear Conditioning, Extinction, Reinstatement, and Fear Testing, all administered in the same apparatus. On day 1, all subjects received 20 min of handling and 20 min of exploration of the apparatus. On day 2, all subjects received two fear conditioning trials, each consisting of a 10 sec 3000 Hz tone (CS), followed by an inescapable .7 sec, 1 mA shock. On day 3, all subjects received 10 fear extinction trials, each a 10 sec 3000 Hz tone (no shock). On day 4, reinstatement was given, consisting of one CS-US pairing (10 sec CS, .7 sec US), in which the CS was either the same (3000 Hz) or different (1000 Hz) tone stimulus to that of conditioning and extinction, depending on the group designation. Fear testing was conducted either on day 5 (Short delay Groups) or day 10 (Long delay Groups). For testing, all subjects received five CS-alone presentations with the 3000 Hz tone (10-sec stimulus presentations, 90-sec ITI), and behavior was videotaped during each tone presentation.
Reinstated fear was measured by freezing responses during the CS on the test day. Freezing was operationally defined as behavior immobility except for respiration movement [23]. For each of the test trials, the rat was judged as freezing or not at 3, 6, and 9 sec of the 10-sec stimulus, and a mean percentage of the freezing measure was calculated for each trial. Coding was performed by two observers blind to the experimental condition, and interrater agreement was 94%.
3. RESULTS AND DISCUSSION
To determine whether an increase in generalization of reinstated fear occurred with time, analyses of variance (ANOVAs) were performed on the percentage of freezing behavior during each CS trial presentation. In the analyses, trials were treated as the repeated measures withing-subjects factor, and Reinstatement CS (Same, Different) and Post-Reinstatement Delay (Short, Long) were between-subjects factors. An alpha level of .05 was used to determine statistical significance. In general, the analyses revealed clear effects of reinstatement stimulus and delay conditions, with support for the notion that reinstated fear increases and generalizes over time.
The mean percentage freezing responses for each group over the five test trials are presented in Fig. (1). These results reveal the effects of both the reinstatement CS and the delay manipulations, as well as an interaction between the two. At a short reinstatement-test interval, greater fear (i.e., more freezing) was observed in the group that received the same CS (Short-Same) than in the group that received a different CS (Short-Diff), suggesting that subjects were able to discriminate between the same and different CS with a short interval between reinstatement and testing. However, with a long delay, this difference was not present. The different reinstatement CS group’s fear level was at a high level, such that there was no significant difference between it and the group that received the same CS (Long-Diff vs. Long-Same). Interestingly, when the same reinstatement CS was used, there appeared to be a fear incubation effect with an increase in fear over time (Short-Same vs. Long-Same).
Statistical analyses supported the following impressions: A 2 (CS) x 2 (Delay) x 5 (Trials) repeated measures ANOVA on freezing percentage (fear) levels revealed a significant delay effect, F(1, 36) = 13.30, p < .01, a significant CS effect, F(1, 36) = 4.39, p < .05, a significant trials effect, F(4, 144) = 2.89, p < .05, and a significant CS x Delay x Trials interaction, F(4, 144) = 3.61, p < .01. No other effects or interactions reached significance. Due to the significant CS x Delay x Trials interaction, separate 2 (CS) x 2 (Delay) ANOVAs were conducted at each trial. These revealed significant effects of CS at Trial 1, F(1,36) = 5.79, and Trial 4, F(1,36) = 4.59, ps < .05, reflecting the greater responding to the same CS than the different CS. There were also significant effects of delay at Trial 1, F(1,36) = 10.94, p < .01, Trial 2, F(1,36) = 9.89, p < .01, Trial 3, F(1,36) = 6.72, p < .05, and Trial 5, F(1,36) = 9.71, p < .01. No interaction effects were significant.
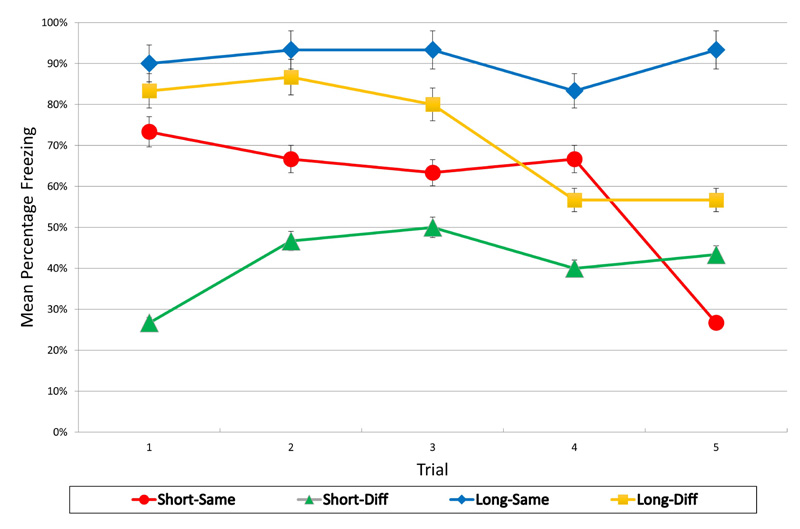
These significant delay effects reflect the overall great fear that occurred with the time delay between reinstatement and testing. For the different reinstatement CS groups, this effect is consistent with the notion of an increase in the generalization of reinstated fear over time. For the same reinstatement CS groups, the observed higher fear levels with a delay after reinstatement are suggestive of a fear incubation effect, a phenomenon not uncommon in the literature [24, 25], and the present study adds to that literature with a demonstration of incubation of reinstated fear to a discrete stimulus.
Experiment 1 demonstrated that after conditioning and extinction, reinstatement with a different stimulus than that used in conditioning and extinction produced lower levels of fear than reinstatement with the same CS, when final fear testing was administered one day after reinstatement Fig. (1), (Groups Short-Same vs. Short-Diff). This suggests that subjects were able to discriminate between the two stimuli. However, when a long delay was implemented between the reinstatement and testing, fear to both stimuli increased. With the different CS, an increase in fear was observed compared to the corresponding short delay group Fig. (1), (Groups Long-Diff vs. Short-Diff), consistent with the notion that reinstated fear generalizes and increases with time. Moreover, for the same CS, the increase in fear observed over time was consistent with an incubation of reinstated CS fear effect (Groups Short-Same vs. Long-Same), something not reported yet in the extant literature on reinstatement and incubation.
A second study was designed to assess whether these fear generalization and incubation effects would be observed if the interval between extinction and reinstatement was delayed instead of the reinstatement-test interval used in Experiment 1. Other than manipulating the delay interval between extinction and reinstatement, all general procedures and dependent measures, as well as the interval between conditioning/extinction and later fear testing, were the same as in the first experiment.
4. METHODS
4.1. Experiment 2
4.1.1. Subjects, Apparatus, and Design
The subjects were 56 rats from the same source as in Experiment 1, and the apparatus was the same. Rats were randomly assigned into groups using a 2 x 2 factorial design, with Post-Extinction Delay (Short, Long) x Reinstatement CS (Same, Diff), yielding 4 groups: Short-Same, Short-Diff, Long-Same, Long-Diff.
4.1.2. Procedure
All general procedures, stimuli, trials, durations, etc., were the same as in Experiment 1. Handling/Exploration, Fear Conditioning, and Extinction treatments occurred on Days 1-3. Reinstatement with either the same or different CS was administered after a short post-extinction delay (Day 4; Groups Short-Same, Short-Diff)) or a long post-extinction delay (Day 9; Groups Long-Same, Long-Diff). Five CS fear testing trials were administered 24 h after the reinstatement treatment (Day 5 or 10), and fear was assessed through freezing behavior during each CS.
5. RESULTS AND DISCUSSION
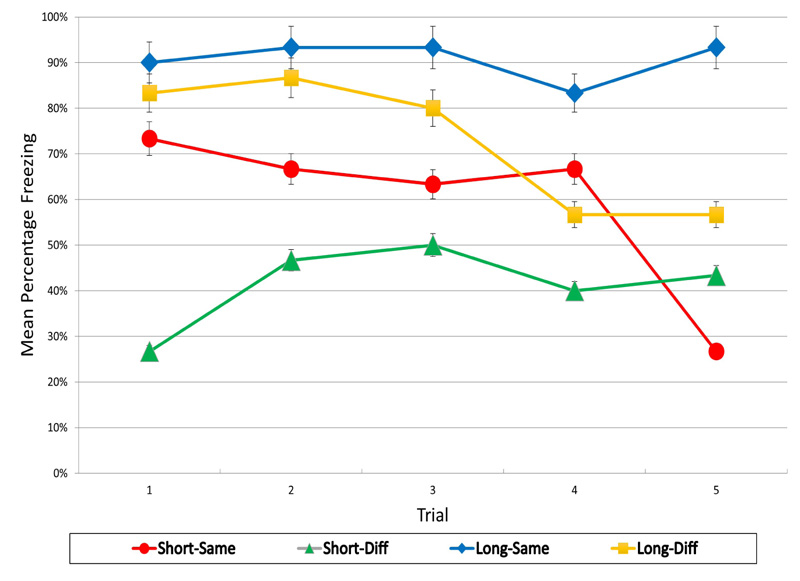
The mean percentage freezing responses for each group over the five test trials are presented in Fig. (2). These results indicate that introducing a delay between extinction and reinstatement produced different effects on reinstated fear than those obtained in Experiment 1 with a post-reinstatement delay. For both the short and long delay conditions, reinstatement with the same CS produced greater levels of fear than reinstatement with a different CS (Same vs. Diff Groups). No increase in generalization of reinstated fear was observed (Group Short-Diff vs. Long-Diff), and no incubation of reinstated fear was observed with the delay (Groups Short-Same vs. Long-Same).
A 2 (CS) x 2 (Delay) x 5 (Trials) repeated measures ANOVA on freezing percentage (fear) levels revealed a significant Trials effect, F(4, 208) = 7.64, p < .01, reflecting the decrease in fear over trials as a function of the CS-alone trial presentations. There was also a significant CS effect, F(1, 52) = 6.48, p < .05, but there was no effect of Delay, F(1, 52) = 0.73, nor any significant interaction effects. The significant CS effect is indicative of the finding that reinstatement with the same CS produced greater fear than with the different CS, regardless of whether the extinction-reinstatement delay was short or long.
CONCLUSION
A primary goal of the present research was to determine if reinstated fear to a CS would generalize and increase over time. To that end, in both experiments, reinstatement was given with the same or different CS than that used in conditioning and extinction, and either the post-reinstatement interval or the post-extinction interval was manipulated. When there was a relatively short interval (1 day) between reinstatement and testing (Exp. 1), reinstatement with a different but similar CS resulted in lower fear than reinstatement with the same CS. However, when the post-reinforcement interval was long (5 days), the resulting fear level with a different reinstatement stimulus was no different than that with the same stimulus, suggesting an increase in generalization of reinstated fear over time (Fig. 1), Groups Long-Diff vs. Long-Same. These findings are similar to other data showing an increased generalization of reinstated context fear over time [16, 21]. The present research adds to the reinstatement literature by demonstrating that reinstated fear generalization effects are not limited to context fear but can also be achieved with discrete stimuli.
The results of Experiment 2 indicated that a critical factor in the increase in reinstated fear generalization is the extinction-reinstatement temporal relationship. When the same delay interval (5 days) used in Experiment 1 occurred post-extinction rather than post-reinstatement, no increase in fear occurred to the different reinstatement stimulus (Fig. 2), (Groups Short-Diff vs. Long-Diff). In both experiments, the testing of final fear occurred after the same interval from conditioning and extinction, yet reinstatement was protected from an increased generalization effect when it was separated for a longer interval from extinction. Taken together, these two experiments suggested that reinstatement can increase fear generalization over time, but there is no increase in generalization of the fear that remains after extinction before any reinstatement.
One unanticipated finding in the first experiment was the greater fear present in the Long-Same group than in the Short-Same group. Reinstatement with the same CS resulted in more fear after a five-day delay than after a one-day delay. This so-called “incubation effect” is certainly not novel in human anxiety research [26] and has been demonstrated with conditioned fear in animals [24, 25] as well as with appetitive conditioned extinction [27]. The results of Experiment 1 are the first to demonstrate incubation of reinstated fear when a long post-reinstatement delay is present. However, as was the case with the increase in generalization of fear effect, this incubation effect again depends on the delay period occurring after reinstatement. No such incubation occurred with a delay after extinction prior to reinstatement (Exp. 2).
These findings have implications for behavior therapies that employ exposure procedures for the treatment of various anxiety disorders. If a client receives exposure treatments to the level where no fear is apparent and shortly after has another traumatic event, even if that trauma is not as intense as the original trauma (reinstated fear was obtained in the present research with a single trial), fear may reappear. If the reinstatement occurs with a different but similar stimulus, that different stimulus may reinstate fear to the original stimulus with time, presumably resulting in more generalization and greater fear the longer time passes. Alternatively, if, after exposure treatments, the client receives another aversive conditioning experience with the same original stimulus, the fear to that reinstated stimulus may increase over time (incubation). Comparing the results of Experiment 2 to Experiment 1, the suggestion is that the more time that elapses after extinction (exposure) treatment before any additional traumatic events is experienced, the lower the chance that fear will either increase through generalization or incubation.
ETHICALS APPROVAL AND CONSENT TO PARTICIPATE
This study was approved by the USC Aiken IACUC (2006 09-P54-02).
HUMAN AND ANIMAL RIGHTS
No humans are used in the studies that are the basis of this research. All animal care and procedures followed were in accordance with the standards set forth in the eighth edition of Guide for the Care and Use of Laboratory Animals,.
CONSENT FOR PUBLICATION
Not applicable.
AVAILABILITY OF DATA AND MATERIALS
The data that support the findings of this study are available from the corresponding author, [EJ.C.], on special request.
FUNDING
The University of South Carolina Aiken, College of Sciences and Psychology Department provided funds for this research.
CONFLICT OF INTEREST
The authors confirm that this article's content has no conflict of interest.
ACKNOWLEDGEMENTS
The authors would like to acknowledge The University of South Carolina Aiken, College of Sciences and Psychology Department, for funding this research. The authors also acknowledge the helpful contributions of Kylie Jupp and Sarah Przywara to this project.


